New reserach published in Acta Neuropathologica Communication
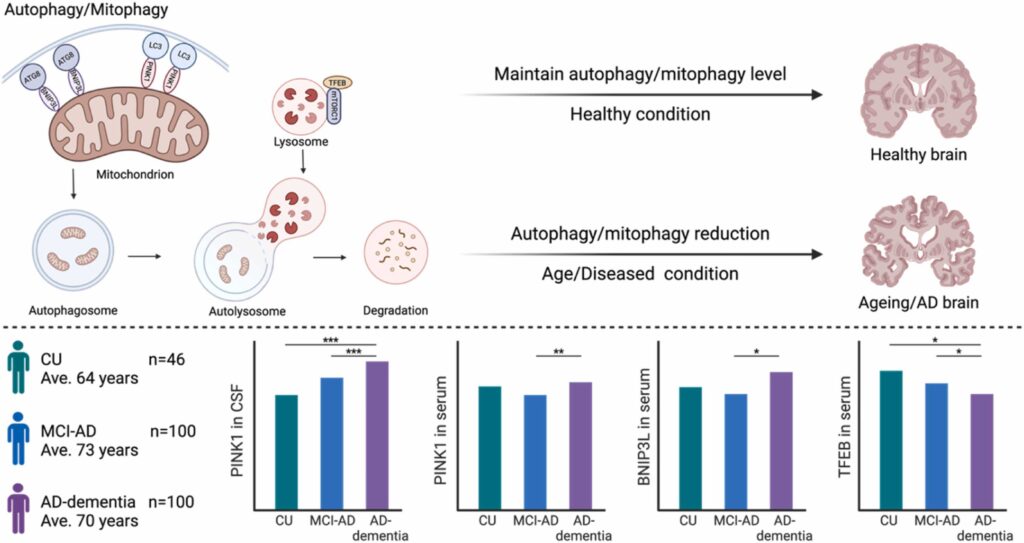
We are pleased to announce the publication of our latest research article, “Distinctive autophagy/mitophagy biomarker profiles in frontotemporal lobar degeneration and Alzheimer’s disease,” in Acta Neuropathologica Communications. This study, published on February 20, 2025, presents our findings on the unique biomarker profiles associated with autophagy and mitophagy in FTLD and AD.
By investigating cerebrospinal fluid (CSF) and serum samples from patients diagnosed with FTLD and AD, both in dementia and MCI stage, this study provides compelling evidence of distinct alterations in autophagy/mitophagy biomarkers between FTLD and AD, indicating that these neurodegenerative diseases may affect the cellular waste disposal system through different pathways. This is the first study to explore mitophagy biomarkers in human CSF and serum in FTLD, opening avenues for further research and potential clinical applications.
We extend our gratitude to our collaborators and funding agencies for their support in this endeavor. For a comprehensive understanding of our findings, we invite you to read the full article at https://actaneurocomms.biomedcentral.com/articles/10.1186/s40478-025-01954-9
Zdroje